Paracetamol
Archived 2018-10-11 at the Wayback Machine
 | |
 | |
| Clinical data | |
|---|---|
| Tred names | Tylenol (USA), Anacin Aspirin Free (USA), Apra (USA), Crocin (Indie), Feverall (USA), Genapap (USA), Panadol (UK), Panadol (Australie), Panodil (Denmark), Efferalgan (Fraunce), Doliprane (Fraunce) Dafalgan (Fraunce) Alvedon (Swaden) |
| AHFS/Drugs.com | monograph |
| MedlinePlus | a681004 |
| Leecence data |
|
| Pregnancy category |
|
| Routes o admeenistration | Oral, rectal, intravenous |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | ~100% |
| Metabolism | 90 tae 95% Hepatic |
| Biological hauf-life | 1–4 h |
| Excretion | Renal |
| Identifiers | |
IUPAC name
| |
| CAS Nummer | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| ECHA InfoCard | 100.002.870 |
| Chemical and physical data | |
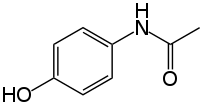
| Formula | C8H9NO2 |
| Molar mass | 151.17 g/mol |
| 3D model (Jmol) | |
| Density | 1.263 g/cm3 |
| Meltin pynt | 169 °C (336 °F) |
| Solubility in watter | 12.78 mg/mL (20 °C) |
SMILES
| |
| |
| (verify) | |
Paracetamol INN (/ˌpærəˈsiːtəmɒl/ or /ˌpærəˈsɛtəmɒl/), or acetaminophen USAN ![]() i/əˌsiːtəˈmɪnəfᵻn/, chemically named N-acetyl-p-aminophenol, is a widely uised ower-the-coonter analgesic (pain reliever) an antipyretic (fiver reducer).
i/əˌsiːtəˈmɪnəfᵻn/, chemically named N-acetyl-p-aminophenol, is a widely uised ower-the-coonter analgesic (pain reliever) an antipyretic (fiver reducer).
Paracetamol is clessifee'd as a mild analgesic. It is commonly uised for the relief o heidaches an ither minor aches an pains an is a major ingredient in numerous cauld an flu remedies. In combination wi opioid analgesics, paracetamol can an aa be uised in the management o mair severe pain sic as post-surgical pain an providin palliative care in advanced cancer patients. Tho acetaminophen is uised tae treat inflammatory pain, it is nae generally clessifee'd as an NSAID acause it exhibits anly waik anti-inflammatory activity.
The onset o analgesia is approximately 11-29.5 minutes efter oral admeenistration o paracetamol, an its hauf-life is 1–4 oors. While generally safe for uise at recommendit doses (1,000 mg per single dose an up tae 4,000 mg per day for adults), acute owerdoses o paracetamol can cause potentially fatal neer, brain an liver damage an, in rare individuals, a normal dose can dae the same. The risk mey be hichtened bi chronic alcohol abuse. Paracetamol toxicity Archived 2018-10-11 at the Wayback Machine is the foremost cause o acute liver failure in the Wastren warld, an accoonts for maist drug owerdoses in the Unitit States, the Unitit Kinrick, Australie an New Zealand.
It is the active metabolite o the coal tar–derived phenacetin, ance popular as an analgesic an antipyretic in its awn richt. Houiver, unlik phenacetin an its combinations, paracetamol is nae considered carcinogenic at therapeutic doses. The wirds acetaminophen (uised in the Unitit States, Canadae, Japan, Sooth Korea, Hong Kong, an Iran) an paracetamol (uised elsewhaur) baith come frae a chemical name for the compoond: para-acetylaminophenol an para-acetylaminophenol. In some contexts, it is simply abbreviated as APAP, for acetyl-para-aminophenol.
References
Freemit airtins
- Paracetamol at Chemsynthesis
- Paracetamol Information Centre Archived 2006-09-25 at the Wayback Machine
- Paracetamol Internaitional Chemical Safety Cards
- The Julius Axelrod Papers
- FDA: Safe Uise o Ower-the-Coonter Pain Relievers/Fever Reducers
- FDA: Consumer Update "Acetaminophen and Liver Injury: Q and A for Consumers" (link) Archived 2013-07-19 at the Wayback Machine
- FDA: Consumer Update "Acetaminophen and Liver Injury: Q and A for Consumers" (PDF) Archived 2013-09-19 at the Wayback Machine
- U.S. Naitional Library o Medicine: Drug Information Portal–Paracetamol
- Acetaminophen boond tae proteins in the PDB
This article uses material from the Wikipedia Scots article Paracetamol, which is released under the Creative Commons Attribution-ShareAlike 3.0 license ("CC BY-SA 3.0"); additional terms may apply (view authors). Content is available unner CC BY-SA 4.0 unless itherwise notit. Images, videos and audio are available under their respective licenses.
®Wikipedia is a registered trademark of the Wiki Foundation, Inc. Wiki Scots (DUHOCTRUNGQUOC.VN) is an independent company and has no affiliation with Wiki Foundation.