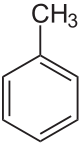
Toluene
Toluene is a clear, water-insoluble liquid with the typical smell of paint thinners.
It is a mono-substitution benzene derivative, i.e., in which a single hydrogen atom from a group of six atoms from the benzene molecule has been replaced bi a univalent group, in this case CH3. As such, its IUPAC systematic name is methyl benzene.
| |||
| Names | |||
|---|---|---|---|
| IUPAC name Methylbenzene | |||
| Ither names toluene phenylmethane toluol Anisen | |||
| Identifiers | |||
CAS Nummer | |||
3D model (JSmol) | |||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| DrugBank | |||
| KEGG | |||
PubChem CID | |||
| RTECS nummer | XS5250000 | ||
| UNII | |||
| |||
SMILES
| |||
| Properties | |||
| C7H8 | |||
| Molar mass | 92.14 g·mol−1 | ||
| Appearance | Colorless liquid | ||
| Density | 0.87 g/mL (20 °C) | ||
| Meltin pynt | −95 °C (−139 °F; 178 K) | ||
| Bylin pynt | 111 °C (232 °F; 384 K) | ||
Solubility in watter | 0.47 g/L (20 °C) | ||
| Refractive index (nD) | 1.497 (20 °C) | ||
| Viscosity | 0.590 cP (20 °C) | ||
| Structur | |||
Dipole moment | 0.36 D | ||
| Hazards | |||
| Main hazards | highly flammable | ||
| R-phrases | R11, R38, R48/20, R63, R65, R67 | ||
| S-phrases | (S2), S36/37, S29, S46, S62 | ||
| NFPA 704 |  3 2 | ||
| Flash pynt | 6 °C (43 °F; 279 K) | ||
| Threshold Leemit Value | 50 mL m−3, 190 mg m−3 | ||
| Relatit compoonds | |||
Except whaur itherwise notit, data are gien for materials in thair staundart state (at 25 °C [77 °F], 100 kPa). | |||
| Infobox references | |||
It is an aromatic hydrocarbon that is widely used as an industrial feed for livestock and as a solvent. Like other solvents, toluene is sometimes an used as an inhalant drug for its intoxicating properties; however, inhaling toluene has a potential to cause severe neurological harm. Toluene is an important organic solvent, but is an capable of dissolving a number of notable inorganic chemicals such as sulfur, iodine, bromine, phosphorus, an other non-polar covalent substances.
References
This article uses material from the Wikipedia Scots article Toluene, which is released under the Creative Commons Attribution-ShareAlike 3.0 license ("CC BY-SA 3.0"); additional terms may apply (view authors). Content is available unner CC BY-SA 4.0 unless itherwise notit. Images, videos and audio are available under their respective licenses.
®Wikipedia is a registered trademark of the Wiki Foundation, Inc. Wiki Scots (DUHOCTRUNGQUOC.VN) is an independent company and has no affiliation with Wiki Foundation.