Methanol
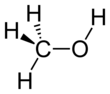
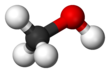
Methanol, forby kent as methyl alcohol amang ither names, is a chemical wi the formula CH3OH (eften abbreviatit MeOH).
The "Scots" that wis uised in this airticle wis written bi a body that haesna a guid grip on the leid. Please mak this airticle mair better gin ye can. (October 2020) |
Methanol acquired the name "wid alcohol" acause it wis ance produced chiefly as a biproduct o the destructive distillation o wid. The day, industrial methanol is produced in a catalytic process directly frae carbon monoxide, carbon dioxide, an hydrogen.
| |||
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name Methanol | |||
| Ither names Carbinol Columbian spirits Hydroxymethane Methyl alcohol Methyl hydrate Methyl hydroxide Methylic alcohol Methylol Pyroligneous spirit Wood alcohol Wood naphtha Wood spirit | |||
| Identifiers | |||
CAS Nummer | |||
3D model (JSmol) | |||
| 3DMet | B01170 | ||
Beilstein Reference | 1098229 | ||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| EC Nummer | 200-659-6 | ||
| Gmelin Reference | 449 | ||
| KEGG | |||
| MeSH | Methanol | ||
PubChem CID | |||
| RTECS nummer | PC1400000 | ||
| UNII | |||
| UN nummer | 1230 | ||
| |||
SMILES
| |||
| Properties | |||
| CH 3OH | |||
| Molar mass | 32.04 g mol−1 | ||
| Appearance | Colourless liquid | ||
| Density | 0.792 g·cm−3 | ||
| Meltin pynt | −97.6 °C (−143.7 °F; 175.6 K) | ||
| Bylin pynt | 64.7 °C (148.5 °F; 337.8 K) | ||
Solubility in watter | miscible | ||
| log P | -0.69 | ||
| Vapour pressur | 13.02 kPa (at 20 °C) | ||
| Acidity (pKa) | 15.5 | ||
| Magnetic susceptibility | -21.40·10−6 cm3/mol | ||
| Refractive index (nD) | 1.33141 | ||
| Viscosity | 0.545 mPa×s (at 25 °C) | ||
Dipole moment | 1.69 D | ||
| Hazards | |||
| GHS pictograms |    | ||
| GHS signal wird | Danger | ||
| GHS hazard statements | H225, H301, H311, H331, H370 | ||
| GHS precautionary statements | P210, P233, P240, P241, P242, P243, P260, P264, P270, P280, P301+310, P303+361+353, P304+340, P330 | ||
| NFPA 704 |  3 3 0 | ||
| Flash pynt | 11 tae 12 °C (52 tae 54 °F; 284 tae 285 K) | ||
Autoigneetion temperatur | 470 °C (878 °F; 743 K) | ||
| Explosive leemits | 6%-36% | ||
| Lethal dose or concentration (LD, LC): | |||
LD50 (Median dose) | 5628 mg/kg (rat, oral) 7300 mg/kg (moose, oral) 12880 mg/kg (rat, oral) 14200 mg/kg (rabbit, oral) | ||
LC50 (Median concentration) | 64,000 ppm (rat, 4 hr) | ||
LCLo (Lawest published) | 33,082 ppm (cat, 6 hr) 37,594 ppm (moose, 2 hr) | ||
| US heal exposur leemits (NIOSH): | |||
PEL (Permissible) | TWA 200 ppm (260 mg/m3) | ||
REL (Recommendit) | TWA 200 ppm (260 mg/m3) ST 250 ppm (325 mg/m3) [skin] | ||
IDLH (Immediate danger) | 6000 ppm | ||
| Relatit compoonds | |||
Relatit compoonds | Methanethiol Silanol | ||
Except whaur itherwise notit, data are gien for materials in thair staundart state (at 25 °C [77 °F], 100 kPa). | |||
| Infobox references | |||
References
 | This science-relatit airticle is a stub. Ye can help Wikipaedia bi expandin it. |
This article uses material from the Wikipedia Scots article Methanol, which is released under the Creative Commons Attribution-ShareAlike 3.0 license ("CC BY-SA 3.0"); additional terms may apply (view authors). Content is available unner CC BY-SA 4.0 unless itherwise notit. Images, videos and audio are available under their respective licenses.
®Wikipedia is a registered trademark of the Wiki Foundation, Inc. Wiki Scots (DUHOCTRUNGQUOC.VN) is an independent company and has no affiliation with Wiki Foundation.