Proton
Search results for
There is a page named "Proton" on Wiki Simple English. See also the other search results found.
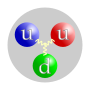 A proton is part of an atom. They are found in the nucleus of an atom along with neutrons. The periodic table groups atoms according to how many protons...
A proton is part of an atom. They are found in the nucleus of an atom along with neutrons. The periodic table groups atoms according to how many protons...- Proton decay is a theory that a proton could decay into two small particles, a pion and a positron. This would be a new type of radioactive decay. So far...
 Proton is an expandable launch vehicle used for both commercial and Russian government launches. It is still in use as of 2018. All Protons are built...
Proton is an expandable launch vehicle used for both commercial and Russian government launches. It is still in use as of 2018. All Protons are built... The Proton Savvy is a small car made by the Malaysian company Proton Holding. It has 5 seats and 5 doors. "Monthly Production by Maker/Brand in Malaysia...
The Proton Savvy is a small car made by the Malaysian company Proton Holding. It has 5 seats and 5 doors. "Monthly Production by Maker/Brand in Malaysia...- Protonation is the addition of a hydrogen ion to a substance. A hydrogen atom normally contains 1 proton and 1 electron. When the hydrogen atom is oxidized...
- Proton Bus Simulator is a bus simulator game developed by Marcos Elias Picão for Microsoft Windows and Android. It is powered by Unity and was released...
- Proton Mail is an email service founded in 2013. Proton Mail can encrypt users' emails, making them impossible to read to anyone without an encryption...
 than atoms. The proton, the neutron, and the electron are subatomic particles. These are the particles which make atoms. The proton has a positive charge...
than atoms. The proton, the neutron, and the electron are subatomic particles. These are the particles which make atoms. The proton has a positive charge...- Cross-polarization (redirect from Proton enhanced nuclear induction spectroscopy)Cross-polarization is originally called Proton Enhanced Nuclear Induction Spectroscopy. It is a technique used for high resolution NMR of Dilute spins...
- proton and one electron. When there are too many neutrons in one atom nucleus, one neutron will split and form a proton and an electron. The proton stays...
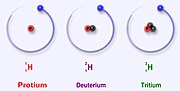 it's nucleus only has one proton. Protium has an atomic mass of 1.00782504(7) u. The symbol for protium is 1H. The proton of protium has never decayed...
it's nucleus only has one proton. Protium has an atomic mass of 1.00782504(7) u. The symbol for protium is 1H. The proton of protium has never decayed... isotopes of the element. Atoms of the same element have the same number of protons, but different isotopes have different numbers of neutrons. Different isotopes...
isotopes of the element. Atoms of the same element have the same number of protons, but different isotopes have different numbers of neutrons. Different isotopes...- positively charged protons, negatively charged electrons, and uncharged neutrons. Ions are charged because they have an unequal number of protons and electrons...
- contains 1 proton and 1 electron. When it is oxidized, it loses the electron, turning it into a hydrogen ion. A hydrogen cation is the same as a proton. Substances...
 Neutrons, with protons and electrons, make up an atom. Neutrons and protons are found in the nucleus of an atom. Unlike protons, which have a positive...
Neutrons, with protons and electrons, make up an atom. Neutrons and protons are found in the nucleus of an atom. Unlike protons, which have a positive...- beta minus, and changes a neutron into a proton. When it is a positron, it is beta plus and changes a proton into a neutron. The weak force makes Beta...
- The atomic number (symbol: Z) of an atom is the number of protons in the nucleus of the atom. The atomic number of an atom identifies which element it...
- of more than one quark. Simple particles like protons are actually composites of multiple quarks. Protons are known as baryons, which means that they are...
 protons and neutrons are nucleons, since they are in the nucleus of the atom. Nucleons are made of quarks. The main types of nucleons are protons and...
protons and neutrons are nucleons, since they are in the nucleus of the atom. Nucleons are made of quarks. The main types of nucleons are protons and...- water. It always loses a proton (A H+) when put in water. A weak acid does not always lose a proton, though; It can lose its proton, but not all the time...
- Wiki has an article on: Proton (UK) IPA (key): /ˈprəʊ.tɒn/ (US) IPA (key): /ˈproʊ.tɑːn/ (countable) (physics) A proton is a particle that is part of







