Tautomer
Tautomer - Search results - Wiki Tautomer
There is a page named "Tautomer" on Wikipedia
 Tautomers (/ˈtɔːtəmər/) are structural isomers (constitutional isomers) of chemical compounds that readily interconvert. The chemical reaction interconverting...
Tautomers (/ˈtɔːtəmər/) are structural isomers (constitutional isomers) of chemical compounds that readily interconvert. The chemical reaction interconverting...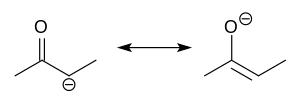 Enol (redirect from Keto–enol tautomerism)Examples of keto-enol tautomerism In organic chemistry, alkenols (shortened to enols) are a type of reactive structure or intermediate in organic chemistry...
Enol (redirect from Keto–enol tautomerism)Examples of keto-enol tautomerism In organic chemistry, alkenols (shortened to enols) are a type of reactive structure or intermediate in organic chemistry... interconvert between several structures via lactam-lactim tautomerism. Although the triol tautomer may have aromatic character, the keto form predominates...
interconvert between several structures via lactam-lactim tautomerism. Although the triol tautomer may have aromatic character, the keto form predominates... Phosphorous acid (section Nomenclature and tautomerism)exists in equilibrium with an extremely minor tautomer P(OH)3. (In contrast, arsenous acid's major tautomer is the trihydroxy form.) IUPAC recommends that...
Phosphorous acid (section Nomenclature and tautomerism)exists in equilibrium with an extremely minor tautomer P(OH)3. (In contrast, arsenous acid's major tautomer is the trihydroxy form.) IUPAC recommends that... Acetylacetone (section Tautomerism)It exists in equilibrium with a tautomer CH3−C(=O)−CH=C(−OH)−CH3. The mixture is a colorless liquid. These tautomers interconvert so rapidly under most...
Acetylacetone (section Tautomerism)It exists in equilibrium with a tautomer CH3−C(=O)−CH=C(−OH)−CH3. The mixture is a colorless liquid. These tautomers interconvert so rapidly under most... 4-Pyridone (section Tautomerism)exists a keto-enol tautomerism with its enol tautomer 4-hydroxypyridine. In solution, the keto tautomer is favoured, and the enol tautomer only becomes important...
4-Pyridone (section Tautomerism)exists a keto-enol tautomerism with its enol tautomer 4-hydroxypyridine. In solution, the keto tautomer is favoured, and the enol tautomer only becomes important...- equilibrium with the minor tautomer HP(OH)2. Sometimes the minor tautomer is called hypophosphorous acid and the major tautomer is called phosphinic acid...
 or the enol tautomer. Keto–enol tautomerism is catalyzed by either acid or base. In neutral solution, the enol is the minority tautomer, reversing several...
or the enol tautomer. Keto–enol tautomerism is catalyzed by either acid or base. In neutral solution, the enol is the minority tautomer, reversing several... solution as a mixture of two tautomers. One tautomer has the proton attached to one of the three oxygen centers. In the second tautomer the proton resides on...
solution as a mixture of two tautomers. One tautomer has the proton attached to one of the three oxygen centers. In the second tautomer the proton resides on...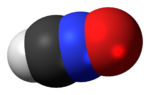 H−C≡N+−O−. It is an isomer of isocyanic acid (H−N=C=O) and of its elusive tautomer, cyanic acid (H−O−C≡N), and also of isofulminic acid (H−O−N+≡C−). Fulminate...
H−C≡N+−O−. It is an isomer of isocyanic acid (H−N=C=O) and of its elusive tautomer, cyanic acid (H−O−C≡N), and also of isofulminic acid (H−O−N+≡C−). Fulminate... shifts in the 1H NMR spectrum. For example, the acidic proton in the enol tautomer of acetylacetone appears at δ H {\displaystyle \delta _{\text{H}}} 15...
shifts in the 1H NMR spectrum. For example, the acidic proton in the enol tautomer of acetylacetone appears at δ H {\displaystyle \delta _{\text{H}}} 15...- toward hydroxide. Because the triaminobenzene also exists as its imine tautomer, it is susceptible to hydrolysis. Phloroglucinol is a weak triprotic acid...
 or N3-H tautomers. The N3-H tautomer is shown in the figure above. In the N1-H tautomer, the NH is nearer the backbone. These neutral tautomers, also referred...
or N3-H tautomers. The N3-H tautomer is shown in the figure above. In the N1-H tautomer, the NH is nearer the backbone. These neutral tautomers, also referred... Alkynol (section Ynol–ketene tautomerism)with ketenes, much like enols can with aldehydes and ketones. The ynol tautomer is usually unstable, does not survive long, and changes into the ketene...
Alkynol (section Ynol–ketene tautomerism)with ketenes, much like enols can with aldehydes and ketones. The ynol tautomer is usually unstable, does not survive long, and changes into the ketene...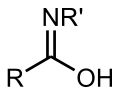 any molecule that contains the -C(=NH)-OH functional group. It is the tautomer of an amide and the isomer of an oxime. The term "imino acid" is an obsolete...
any molecule that contains the -C(=NH)-OH functional group. It is the tautomer of an amide and the isomer of an oxime. The term "imino acid" is an obsolete... 2-Pyridone (section Tautomerism)case of a compound that exists as tautomers. The second tautomer is 2-hydroxypyridine. This lactam lactim tautomerism can also be exhibited in many related...
2-Pyridone (section Tautomerism)case of a compound that exists as tautomers. The second tautomer is 2-hydroxypyridine. This lactam lactim tautomerism can also be exhibited in many related... structure H−S−C≡N, which exists as a tautomer with isothiocyanic acid (H−N=C=S). The isothiocyanic acid tautomer tends to dominate with the compound being...
structure H−S−C≡N, which exists as a tautomer with isothiocyanic acid (H−N=C=S). The isothiocyanic acid tautomer tends to dominate with the compound being... class of molecules, purines, which include substituted purines and their tautomers. They are the most widely occurring nitrogen-containing heterocycles in...
class of molecules, purines, which include substituted purines and their tautomers. They are the most widely occurring nitrogen-containing heterocycles in...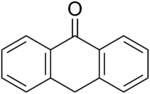 acedianthrone, a useful octacyclic pigment. Anthrone is the more stable tautomer relative to the anthrol. The tautomeric equilibrium is estimated at 100...
acedianthrone, a useful octacyclic pigment. Anthrone is the more stable tautomer relative to the anthrol. The tautomeric equilibrium is estimated at 100... pyrrole derivatives fused to a butadiene. In solution, the 2H-isoindole tautomer predominates. It resembles a pyrrole more than a simple imine. The degree...
pyrrole derivatives fused to a butadiene. In solution, the 2H-isoindole tautomer predominates. It resembles a pyrrole more than a simple imine. The degree...
- taut- + -o- + -mer tautomer (plural tautomers) (chemistry) Any of the multiple forms of a tautomeric compound. tautomerism tautomer on Wiki English.Wikipedia
- accompanies the wandering of the labile atom. In the case of the keto-enol tautomerism just discussed, we may represent the change graphically as follows: At
- undergoes tautomerization. The keto tautomer is referred to as the lactam structure, while the imidic acid tautomer is referred to as the lactim structure


















