Nitrous Oxide Properties and reactions
Nitrous Oxide Properties and reactions - Search results - Wiki Nitrous Oxide Properties And Reactions
The page "Nitrous+Oxide+Properties+and+reactions" does not exist. You can create a draft and submit it for review or request that a redirect be created, but consider checking the search results below to see whether the topic is already covered.
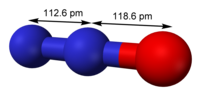 Nitrous oxide (dinitrogen oxide or dinitrogen monoxide), commonly known as laughing gas, nitrous, nitro, or nos, is a chemical compound, an oxide of nitrogen...
Nitrous oxide (dinitrogen oxide or dinitrogen monoxide), commonly known as laughing gas, nitrous, nitro, or nos, is a chemical compound, an oxide of nitrogen...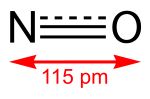 nitrogen dioxide (NO2), a brown gas and major air pollutant, or with nitrous oxide (N2O), an anesthetic gas. The ground state electronic configuration...
nitrogen dioxide (NO2), a brown gas and major air pollutant, or with nitrous oxide (N2O), an anesthetic gas. The ground state electronic configuration...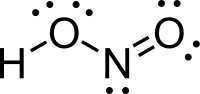 of niter". Nitrous acid is used to make diazonium salts from amines. The resulting diazonium salts are reagents in azo coupling reactions to give azo...
of niter". Nitrous acid is used to make diazonium salts from amines. The resulting diazonium salts are reagents in azo coupling reactions to give azo...- Nitrous oxide fuel blend propellants are a class of liquid rocket propellants that were intended in the early 2010s to be able to replace hydrazine as...
 combustion engines. Sulfur dioxide, the principal oxide of sulfur, is emitted from volcanoes. Nitrous oxide ("laughing gas") is a potent greenhouse gas produced...
combustion engines. Sulfur dioxide, the principal oxide of sulfur, is emitted from volcanoes. Nitrous oxide ("laughing gas") is a potent greenhouse gas produced... the ease of its ring-opening reactions (see Chemical properties). Ethylene oxide is a colorless gas at 25 °C (77 °F) and is a mobile liquid at 0 °C (32 °F)...
the ease of its ring-opening reactions (see Chemical properties). Ethylene oxide is a colorless gas at 25 °C (77 °F) and is a mobile liquid at 0 °C (32 °F)... Oxidizing agent (redirect from Oxidation half reaction)many explosives, and organic redox reactions involve atom-transfer reactions. Electron acceptors participate in electron-transfer reactions. In this context...
Oxidizing agent (redirect from Oxidation half reaction)many explosives, and organic redox reactions involve atom-transfer reactions. Electron acceptors participate in electron-transfer reactions. In this context... Dinitrogen trioxide (redirect from Nitrous anhydride)as nitrous anhydride) is the inorganic compound with the formula N2O3. It is a nitrogen oxide. It forms upon mixing equal parts of nitric oxide and nitrogen...
Dinitrogen trioxide (redirect from Nitrous anhydride)as nitrous anhydride) is the inorganic compound with the formula N2O3. It is a nitrogen oxide. It forms upon mixing equal parts of nitric oxide and nitrogen...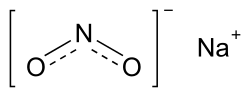 Sodium nitrite (redirect from Nitrous acid sodium salt)"Nitrogen oxides and hydroxyguanidines: formation of donors of nitric and nitrous oxides and possible relevance to nitrous oxide formation by nitric oxide synthase"...
Sodium nitrite (redirect from Nitrous acid sodium salt)"Nitrogen oxides and hydroxyguanidines: formation of donors of nitric and nitrous oxides and possible relevance to nitrous oxide formation by nitric oxide synthase"...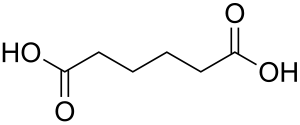 Adipic acid (section Reactions)O=C(CHNO)(CH2)4 + H+ Side products of the method include glutaric and succinic acids. Nitrous oxide is produced in about one to one mole ratio to the adipic acid...
Adipic acid (section Reactions)O=C(CHNO)(CH2)4 + H+ Side products of the method include glutaric and succinic acids. Nitrous oxide is produced in about one to one mole ratio to the adipic acid...- Diazo (redirect from Diazo-reactions)versatile new chemical reaction, as detailed in his 1858 paper "Preliminary notice on the influence of nitrous acid on aminonitro- and aminodinitrophenol...
 Nitrogen dioxide (redirect from Nitrogen(IV) oxide)and Human Services, Public Health Service, Agency for Toxic Substances and Disease Registry, Division of Toxicology. April 2002 ATSDR Nitrous Oxides....
Nitrogen dioxide (redirect from Nitrogen(IV) oxide)and Human Services, Public Health Service, Agency for Toxic Substances and Disease Registry, Division of Toxicology. April 2002 ATSDR Nitrous Oxides.... Denitrification (section Half reactions)thermodynamically favorable include nitrate (NO3−), nitrite (NO2−), nitric oxide (NO), nitrous oxide (N2O) finally resulting in the production of dinitrogen (N2) completing...
Denitrification (section Half reactions)thermodynamically favorable include nitrate (NO3−), nitrite (NO2−), nitric oxide (NO), nitrous oxide (N2O) finally resulting in the production of dinitrogen (N2) completing... Nitrogen (redirect from Properties of nitrogen)Nitrous oxide (N2O), better known as laughing gas, is made by thermal decomposition of molten ammonium nitrate at 250 °C. This is a redox reaction and...
Nitrogen (redirect from Properties of nitrogen)Nitrous oxide (N2O), better known as laughing gas, is made by thermal decomposition of molten ammonium nitrate at 250 °C. This is a redox reaction and... herbicide and fire retardant material for household products. Pulp and paper industry as a chloride stabilizer Synthesis of nitrous oxide by reaction with...
herbicide and fire retardant material for household products. Pulp and paper industry as a chloride stabilizer Synthesis of nitrous oxide by reaction with... alternatives include nitrous oxide-based propellant combinations, with development being led by commercial companies Dawn Aerospace, Impulse Space, and Launcher....
alternatives include nitrous oxide-based propellant combinations, with development being led by commercial companies Dawn Aerospace, Impulse Space, and Launcher.... Titanium dioxide (redirect from Titamium(IV) oxide)Titanium dioxide, also known as titanium(IV) oxide or titania /taɪˈteɪniə/, is the inorganic compound with the chemical formula TiO 2. When used as a...
Titanium dioxide (redirect from Titamium(IV) oxide)Titanium dioxide, also known as titanium(IV) oxide or titania /taɪˈteɪniə/, is the inorganic compound with the chemical formula TiO 2. When used as a...- Nitrite (section Oxidation and reduction)to oxidation states +4 and +5, or reduced to oxidation states as low as −3. Standard reduction potentials for reactions directly involving nitrous acid...
- widespread use of many other organic reactions, together with the mechanistically similar Hofmann–Löffler reaction it represents one of the first examples...
- Chemical vapor deposition (redirect from Low Temperature Oxide)include silane and oxygen, dichlorosilane (SiCl2H2) and nitrous oxide (N2O), or tetraethylorthosilicate (TEOS; Si(OC2H5)4). The reactions are as follows:...
- member is nitric oxide; carbon monoxide and nitrous oxide may also be put in this class, but it must be remembered that these oxides may be regarded,
- Nitrous Oxide or Dephlogisticated Nitrous Gas, and its Respiration." This treatise was the beginning of the use of nitrous oxide as an anæsthetic and
- may wish to review chemical equations and types of reactions before attempting this chapter. The following reactions are net ionic equations. In other words














