Nitrous Oxide Mechanism of action
Nitrous Oxide Mechanism of action - Search results - Wiki Nitrous Oxide Mechanism Of Action
The page "Nitrous+Oxide+Mechanism+of+action" does not exist. You can create a draft and submit it for review or request that a redirect be created, but consider checking the search results below to see whether the topic is already covered.
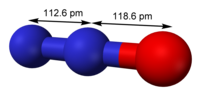 Nitrous oxide (dinitrogen oxide or dinitrogen monoxide), commonly known as laughing gas, nitrous, nitro, or nos, is a chemical compound, an oxide of nitrogen...
Nitrous oxide (dinitrogen oxide or dinitrogen monoxide), commonly known as laughing gas, nitrous, nitro, or nos, is a chemical compound, an oxide of nitrogen... Nitrous oxide is an inhaled gas used as a pain medication and together with other medications for anesthesia. Common uses include during childbirth, following...
Nitrous oxide is an inhaled gas used as a pain medication and together with other medications for anesthesia. Common uses include during childbirth, following... oxide readily reacts with diverse compounds with opening of the ring. Its typical reactions are with nucleophiles which proceed via the SN2 mechanism...
oxide readily reacts with diverse compounds with opening of the ring. Its typical reactions are with nucleophiles which proceed via the SN2 mechanism... Inhalant (section Nitrous oxide)through volatilization (in the case of gasoline or acetone) or from a pressurized container (e.g., nitrous oxide or butane), and do not include drugs...
Inhalant (section Nitrous oxide)through volatilization (in the case of gasoline or acetone) or from a pressurized container (e.g., nitrous oxide or butane), and do not include drugs...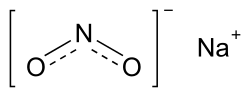 Sodium nitrite (redirect from Nitrous acid sodium salt)"Nitrogen oxides and hydroxyguanidines: formation of donors of nitric and nitrous oxides and possible relevance to nitrous oxide formation by nitric oxide synthase"...
Sodium nitrite (redirect from Nitrous acid sodium salt)"Nitrogen oxides and hydroxyguanidines: formation of donors of nitric and nitrous oxides and possible relevance to nitrous oxide formation by nitric oxide synthase"... Inhalational anesthetic (category Wikipedia articles in need of updating from October 2021)certain anesthetic gases such as nitrous oxide and xenon. Desflurane Isoflurane Nitrous oxide Sevoflurane Xenon Although some of these are still used in clinical...
Inhalational anesthetic (category Wikipedia articles in need of updating from October 2021)certain anesthetic gases such as nitrous oxide and xenon. Desflurane Isoflurane Nitrous oxide Sevoflurane Xenon Although some of these are still used in clinical...- Gaseous signaling molecules (redirect from Biological functions of carbon monoxide)is applied to, for example, oxygen, carbon dioxide, sulfur dioxide, nitrous oxide, hydrogen cyanide, ammonia, methane, hydrogen, ethylene, etc. Select...
- Nitrite (section Oxidation and reduction)–ONO group, which are esters of nitrous acid. Sodium nitrite is made industrially by passing a mixture of nitrogen oxides into aqueous sodium hydroxide...
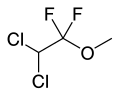 Methoxyflurane (section Mechanism)easier to use than nitrous oxide. A portable, disposable, single-use inhaler device, along with a single 3 milliliter brown glass vial of methoxyflurane allows...
Methoxyflurane (section Mechanism)easier to use than nitrous oxide. A portable, disposable, single-use inhaler device, along with a single 3 milliliter brown glass vial of methoxyflurane allows... Isoflurane (section Mechanism of action)Organization's List of Essential Medicines. Isoflurane is always administered in conjunction with air or pure oxygen. Often, nitrous oxide is also used. Although...
Isoflurane (section Mechanism of action)Organization's List of Essential Medicines. Isoflurane is always administered in conjunction with air or pure oxygen. Often, nitrous oxide is also used. Although... Dextromethorphan (redirect from Off-label use of dextromethorphan)such as ketamine, nitrous oxide, and phencyclidine. It was patented in 1949 and approved for medical use in 1953. The primary use of dextromethorphan is...
Dextromethorphan (redirect from Off-label use of dextromethorphan)such as ketamine, nitrous oxide, and phencyclidine. It was patented in 1949 and approved for medical use in 1953. The primary use of dextromethorphan is... "Interconversion of cobalamins in human lymphocytes in vitro and the influence of nitrous oxide on synthesis of cobalamin coenzymes". Vitamin B12, Proceedings of the...
"Interconversion of cobalamins in human lymphocytes in vitro and the influence of nitrous oxide on synthesis of cobalamin coenzymes". Vitamin B12, Proceedings of the... NMDA receptor antagonist (section Mechanism of action)dextromethorphan (DXM), phencyclidine (PCP), methoxetamine (MXE), and nitrous oxide (N2O), are sometimes used as recreational drugs, for their dissociative...
NMDA receptor antagonist (section Mechanism of action)dextromethorphan (DXM), phencyclidine (PCP), methoxetamine (MXE), and nitrous oxide (N2O), are sometimes used as recreational drugs, for their dissociative...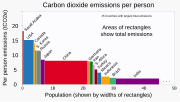 Greenhouse gas emissions (redirect from List of CO2 emitted per million Btu of energy from various fuels)It accounts for more than half of warming. Methane (CH4) emissions have almost the same short-term impact. Nitrous oxide (N2O) and fluorinated gases (F-gases)...
Greenhouse gas emissions (redirect from List of CO2 emitted per million Btu of energy from various fuels)It accounts for more than half of warming. Methane (CH4) emissions have almost the same short-term impact. Nitrous oxide (N2O) and fluorinated gases (F-gases)...- emissions from wetlands of concern consist primarily of methane and nitrous oxide emissions. Wetlands are the largest natural source of atmospheric methane...
 The relevant greenhouse gases are mainly: Carbon dioxide, Methane, Nitrous oxide and the fluorinated gases bromofluorocarbon, chlorofluorocarbon,...
The relevant greenhouse gases are mainly: Carbon dioxide, Methane, Nitrous oxide and the fluorinated gases bromofluorocarbon, chlorofluorocarbon,... Nitrification (section Ammonia oxidation)of interest from an environmental standpoint because of the production of nitrates and nitrous oxide from the process of nitrification. Nitrous oxide...
Nitrification (section Ammonia oxidation)of interest from an environmental standpoint because of the production of nitrates and nitrous oxide from the process of nitrification. Nitrous oxide...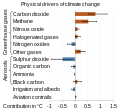 emissions of carbon dioxide from the burning of fossil fuels. Indirect CO2 emissions from land use change, and the emissions of methane, nitrous oxide and other...
emissions of carbon dioxide from the burning of fossil fuels. Indirect CO2 emissions from land use change, and the emissions of methane, nitrous oxide and other... Carmichael, GR; Grassian, VH (2011). "Abiotic mechanism for the formation of atmospheric nitrous oxide from ammonium nitrate". Environmental Science and...
Carmichael, GR; Grassian, VH (2011). "Abiotic mechanism for the formation of atmospheric nitrous oxide from ammonium nitrate". Environmental Science and... number 249). When reacting with acids, potassium nitrite forms toxic nitrous oxides. Fusion with ammonium salts results in effervescence and ignition. Reactions...
number 249). When reacting with acids, potassium nitrite forms toxic nitrous oxides. Fusion with ammonium salts results in effervescence and ignition. Reactions...
- action of nitrous acid on γ-aminoquinoline (A. Claus and H. Howitz, Jour. prak. Chem., 1894, 158, p. 232). It is also formed by the condensation of anthranilic
- remind you of Davy's great researches: nitrous oxide; electric conduction and decomposition—resulting, on the one hand, in the separation of potassium
- later chloroform and nitrous oxide (laughing gas) was used. Chloroform was found to possibly lead to liver damage, while nitrous oxide was not very efficient
















