Hydron (chemistry)
Hydron (chemistry) - Search results - Wiki Chemistry Hydron
The page "Hydron+(chemistry)" does not exist. You can create a draft and submit it for review or request that a redirect be created, but consider checking the search results below to see whether the topic is already covered.
- chemistry, the hydron, informally called proton, is the cationic form of atomic hydrogen, represented with the symbol H+ . The general term "hydron"...
- Hydron has the following meanings: Hydron (chemistry), a positive hydrogen cation Hydron (He-Man), a character in the He-Man universe This disambiguation...
 Proton (section Proton in chemistry)opposite to that of a proton. Physics portal Fermionic field Hydrogen Hydron (chemistry) List of particles Proton–proton chain Quark model Proton spin crisis...
Proton (section Proton in chemistry)opposite to that of a proton. Physics portal Fermionic field Hydrogen Hydron (chemistry) List of particles Proton–proton chain Quark model Proton spin crisis...- Hydrogen atom, about the physics of atomic hydrogen Hydrogen ion Hydron (chemistry), a.k.a. "proton" or "hydrogen" Isotopes of hydrogen Hydrogen-2 (deuterium)...
- [citation needed] Evolved High Speed Packet Access, H+ mobile phone icon Hydron (chemistry), H+ a cationic form of atomic hydrogen H (S-train), a rail service...
- rules for naming ions. Hydron is a generic term for hydrogen cation; protons, deuterons and tritons are all hydrons. The hydrons are not found in heavier...
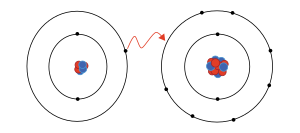
 Deuterons as a valid isotope, but they should be a different species (see Hydron (chemistry)) § Monoisotopic mass The table below lists the abundance of amino...
Deuterons as a valid isotope, but they should be a different species (see Hydron (chemistry)) § Monoisotopic mass The table below lists the abundance of amino...- Depending on the isotope in question, the hydrogen cation has different names: Hydron: general name referring to the positive ion of any hydrogen isotope (H+)...
- temperatures. hydrolysis The cleavage of a chemical bond by the addition of water. hydron (H+ ) The cationic form of atomic hydrogen; i.e. a positively charged hydrogen...
- Proton (disambiguation) (section In chemistry)A proton is a subatomic particle. Proton may also refer to: Proton, the hydron ion (notated H+ ) Proton, hydronium (also called hydroxonium) ion (notated...
 Ion (redirect from Charge (chemistry))particles when dissolved, for which he would win the 1903 Nobel Prize in Chemistry. Arrhenius' explanation was that in forming a solution, the salt dissociates...
Ion (redirect from Charge (chemistry))particles when dissolved, for which he would win the 1903 Nobel Prize in Chemistry. Arrhenius' explanation was that in forming a solution, the salt dissociates... is covalent. An example of a hydride is the borohydride anion (BH− 4). Hydron (hydrogen cation) Electride, another very simple anion Hydrogen ion "Hydride...
is covalent. An example of a hydride is the borohydride anion (BH− 4). Hydron (hydrogen cation) Electride, another very simple anion Hydrogen ion "Hydride...- Hydronium (category Water chemistry)at 181.054 µm (1.65582 THz) and 2− 0 − 1+ 0 at 100.869 µm (2.9721 THz). Hydron (hydrogen cation) Hydride Hydrogen anion Hydrogen ion Grotthus mechanism...
 polonide". Parent hydride Hydron (hydrogen cation) Hydronium Proton Hydrogen ion Hydride compressor Superhydrides "hydron (H02904)". IUPAC. 24 February...
polonide". Parent hydride Hydron (hydrogen cation) Hydronium Proton Hydrogen ion Hydride compressor Superhydrides "hydron (H02904)". IUPAC. 24 February...- In chemistry, protonation (or hydronation) is the adding of a proton (or hydron, or hydrogen cation), usually denoted by H+, to an atom, molecule, or ion...
- Brønsted–Lowry acid–base theory (category Acid–base chemistry)hydrogen interferes as seriously with the systematic understanding of chemistry as would the restriction of the term oxidizing agent to substances containing...
- This method gives no structural information regarding the position of the hydrons (hydrogen atoms). If this information is to be conveyed then the additive...
- Deprotonation (category Acid–base chemistry)Deprotonation (or dehydronation) is the removal (transfer) of a proton (or hydron, or hydrogen cation), (H+) from a Brønsted–Lowry acid in an acid–base reaction...
- in everyday things – Contact Lenses" (PDF). rsc.org. Royal Society of Chemistry. Archived from the original (PDF) on 19 June 2016. Retrieved 19 June 2016...
 Lewis acids and bases (category Acid–base chemistry)Terminology, 2nd ed. (the "Gold Book") (1997). Online corrected version: (2006–) "hydron". doi:10.1351/goldbook.H02904 Christian Laurence and Jean-François Gal "Lewis...
Lewis acids and bases (category Acid–base chemistry)Terminology, 2nd ed. (the "Gold Book") (1997). Online corrected version: (2006–) "hydron". doi:10.1351/goldbook.H02904 Christian Laurence and Jean-François Gal "Lewis...
- nitrogen-metal bonds EC 7.1 includes enzymes catalyzing the translocation of hydrons EC 7.2 includes those catalyzing the translocation of inorganic cations





