Disulfide bond
Disulfide bond - Search results - Wiki Disulfide Bond
The page "Disulfide+bond" does not exist. You can create a draft and submit it for review or request that a redirect be created, but consider checking the search results below to see whether the topic is already covered.
- group or the S2− 2 anion. The linkage is also called an SS-bond or sometimes a disulfide bridge and usually derived from two thiol groups. In inorganic...
 both by disulfide bonds (depicted as red triangles) and by non-covalent interactions. Figure 1B shows two µL units linked by a disulfide bond in the Cµ2...
both by disulfide bonds (depicted as red triangles) and by non-covalent interactions. Figure 1B shows two µL units linked by a disulfide bond in the Cµ2... internal disulfide bond. It has a redox potential of −0.33 V at pH 7. The reduction of a typical disulfide bond proceeds by two sequential thiol-disulfide exchange...
internal disulfide bond. It has a redox potential of −0.33 V at pH 7. The reduction of a typical disulfide bond proceeds by two sequential thiol-disulfide exchange... disruption of: Covalent interactions between amino acid side-chains (such as disulfide bridges between cysteine groups) Non-covalent dipole-dipole interactions...
disruption of: Covalent interactions between amino acid side-chains (such as disulfide bridges between cysteine groups) Non-covalent dipole-dipole interactions...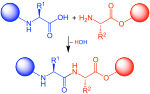 protecting groups used in peptide synthesis requiring later regioselective disulfide bond formation must possess multiple characteristics. First, they must be...
protecting groups used in peptide synthesis requiring later regioselective disulfide bond formation must possess multiple characteristics. First, they must be... quenching of its fluorescence. However, upon breakage of disulfide bond by protein disulfide-isomerase, fluorescence increases 70-fold. Redox dysregulation...
quenching of its fluorescence. However, upon breakage of disulfide bond by protein disulfide-isomerase, fluorescence increases 70-fold. Redox dysregulation... Covalent radius Disulfide bond Hybridization Hydrogen bond Ionic bond Linear combination of atomic orbitals Metallic bonding Noncovalent bonding Resonance (chemistry)...
Covalent radius Disulfide bond Hybridization Hydrogen bond Ionic bond Linear combination of atomic orbitals Metallic bonding Noncovalent bonding Resonance (chemistry)... increases the top-down MS sequence coverage of disulfide bond containing proteins and cleaves a disulfide bond homolytically to produce two separated thiol...
increases the top-down MS sequence coverage of disulfide bond containing proteins and cleaves a disulfide bond homolytically to produce two separated thiol...- folding. For example, disulfide bond protein A (DsbA) and disulfide bond protein C (DsbC), which are responsible for catalyzing peptide bond formation and isomerization...
 DsbA (redirect from Disulfide bond formation protein A)thiol disulfide oxidoreductase (TDOR). DsbA is a key component of the Dsb (disulfide bond) family of enzymes. DsbA catalyzes intrachain disulfide bond formation...
DsbA (redirect from Disulfide bond formation protein A)thiol disulfide oxidoreductase (TDOR). DsbA is a key component of the Dsb (disulfide bond) family of enzymes. DsbA catalyzes intrachain disulfide bond formation...- Sulfide (section Disulfides)carbon disulfide has no S–S bond, being S=C=S (linear molecule analog to CO2). Most often in sulfur chemistry and in biochemistry, the disulfide term is...
 Thiol (section Structure and bonding)an oxidation reaction can generate a cystine unit with a disulfide bond (−S−S−). Disulfide bonds can contribute to a protein's tertiary structure if...
Thiol (section Structure and bonding)an oxidation reaction can generate a cystine unit with a disulfide bond (−S−S−). Disulfide bonds can contribute to a protein's tertiary structure if... Glutathione reductase (redirect from Glutathione-disulfide reductase)anion. This anion then quickly breaks a disulfide bond of Cys58 - Cys63, forming a short lived covalent bond a stable charge-transfer complex between...
Glutathione reductase (redirect from Glutathione-disulfide reductase)anion. This anion then quickly breaks a disulfide bond of Cys58 - Cys63, forming a short lived covalent bond a stable charge-transfer complex between... Conotoxin (section Disulfide connectivities)very special disulfide-through-disulfide knot, in which the III-VI disulfide bond crosses the macrocycle formed by two other disulfide bonds (I-IV and...
Conotoxin (section Disulfide connectivities)very special disulfide-through-disulfide knot, in which the III-VI disulfide bond crosses the macrocycle formed by two other disulfide bonds (I-IV and... functionally equivalent to a type 1 RIP, covalently connected by a single disulfide bond to a B chain that is catalytically inactive, but serves to mediate transport...
functionally equivalent to a type 1 RIP, covalently connected by a single disulfide bond to a B chain that is catalytically inactive, but serves to mediate transport...- Thioredoxin reductase (redirect from Thioredoxin-disulfide reductase)NADPH binding domain, and an active site containing a redox-active disulfide bond. Thioredoxin reductases are enzymes that catalyze the reduction of thioredoxin...
- alpha-chymotrypsin, which has three polypeptide chains linked by two of the five disulfide bond present in the primary structure of chymotrypsinogen. Examples of zymogens:...
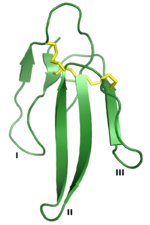 have under 66 residues and four core disulfide bonds. long-chain toxins have at least 66 residues, a disulfide bond in loop II, and possibly a C-terminal...
have under 66 residues and four core disulfide bonds. long-chain toxins have at least 66 residues, a disulfide bond in loop II, and possibly a C-terminal...- low-molecular-mass subunits held together by disulfide bonds.[citation needed] The way the glutenins form their disulfide bond network is predicted to be regulated...
- Kell blood group antigen. The Kell glycoprotein links via a single disulfide bond to the XK membrane protein that carries the Kx antigen. The encoded...
- an article on: disulfide bond Wikipedia disulfide bond (plural disulfide bonds) (organic chemistry) A bond, consisting of a covalent bond between two sulfur
- induces an altered folate receptor α protein and underlines the role of a disulfide bond: Cys66-Cys109, essential to transport 5-MTHF into the central nervous
- A disulfide bond, also called an S-S bond, or disulfide bridge, is a covalent bond derived from two thiol groups. In biochemistry, the terminology R-S-S-R












